
In fact, RPPA protocols report using western blotting as the method for validating antibodies for RPPA use 22. Reverse phase protein array (RPPA) is a well-established method for quantitative data from cell and tissue lysates 19, 20, 21, 22, but it does not separate proteins by molecular weight, and therefore has more stringent requirements for antibody validity. While the western blot is often considered semi-quantitative or qualitative, it can be quantitative with infrared fluorescence-based detection 13, 14, 15, 16, 17, 18. One major application of antibodies in both large and small labs is the western blot. Antibody validity is highly dependent on biological context and the assay itself 10, 11, 12. 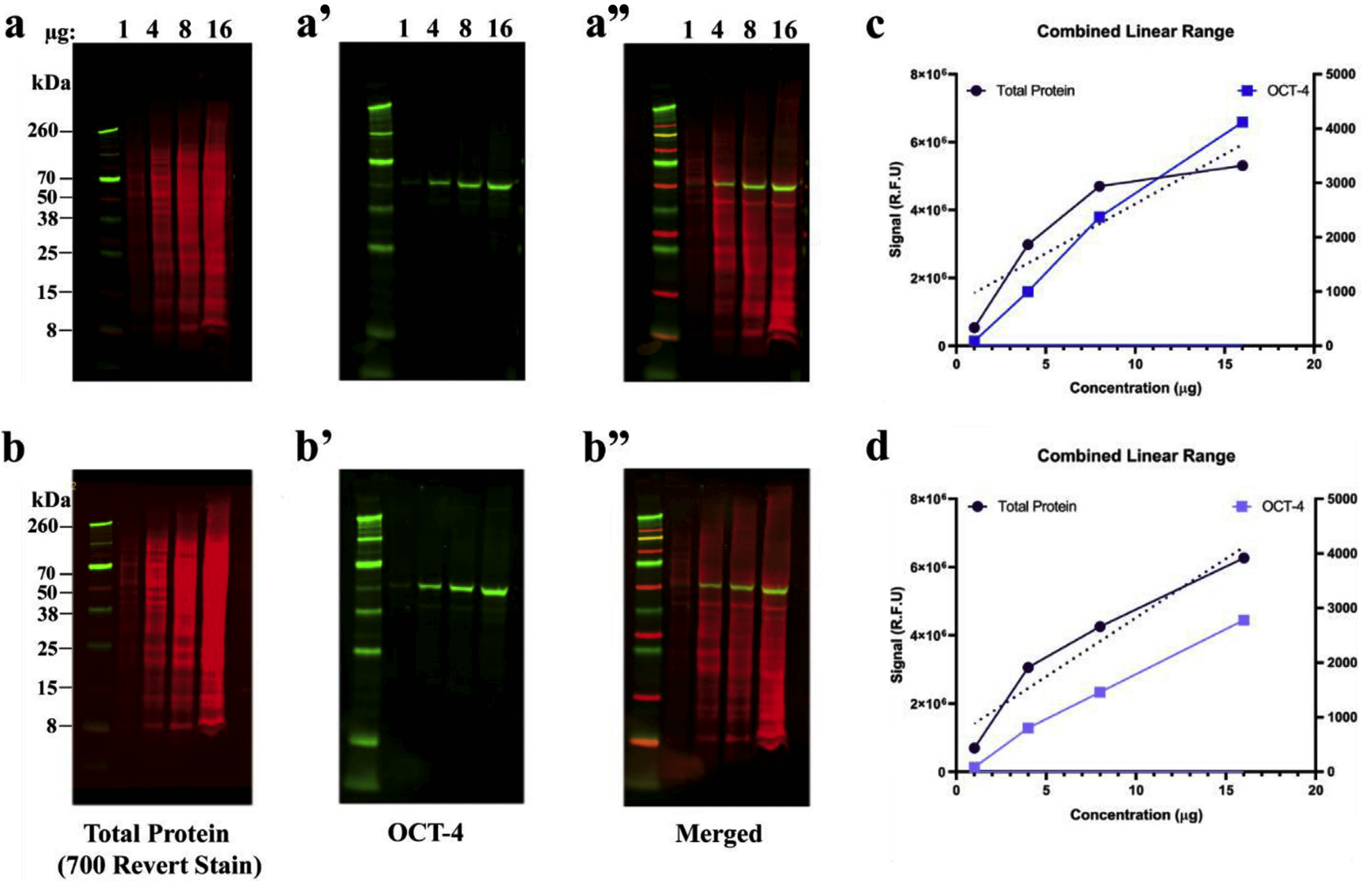
For example, qualitative inference from immunohistochemistry may be possible, but acquiring quantitative data from flow cytometry may not be with the same antibody and cell system. The intended application for the antibody brings potentially different criteria and stringency for their use. Antibodies are widely-used critical tools in a variety of biomedical research assays, but they are not always suitable for the application of interest. While there are many potential reasons for lack of reproducibility, one major reason relates to research reagents, including antibodies 10, 11, 12. Scientific research, and in particular that in the biomedical field, has come under harsh scrutiny and debate of late due to questions of reproducibility 1, 2, 3, 4, 5, 6, 7, 8, 9. Importantly, we demonstrate that results from microwestern analyses scale to normal “macro” western for a subset of antibodies. Total antibodies generally required higher working concentrations, but more comprehensive antibody panels are required to better establish whether this trend is general or not. Phospho-specific and total antibodies do not have discernable trend differences in linear range or limit of detection. Linear range for all validated antibodies is at least 8-fold, and up to two orders of magnitude. In some cases microwestern struggled with higher molecular weight membrane proteins, so the technique may not be uniformly applicable to all validation tasks. Pilot experiments demonstrate a high proportion of investigated antibodies (17/24) are suitable for quantitative use however this sample of antibodies is not yet comprehensive across companies, molecular weights, and other important antibody properties, so the ubiquity of this property cannot yet be determined. Here, we use microwestern array to rapidly evaluate suitable conditions for quantitative western blotting, with up to 192 antibody/dilution/replicate combinations on a single standard size gel with a seven-point, two-fold lysate dilution series (~100-fold range). Fluorescence-based western blots are quantitative in principal, but require determining linear range for each antibody.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
